MEDICUS engineering ApS
Founded in 2001 as a research and development company
Focuses entirely on development, production and sales of medical devices
MEDICUS engineering’s quality management system complies with ISO 13485
MEDICUS engineering’s product portfolio includes:
A new game changing diagnostic device
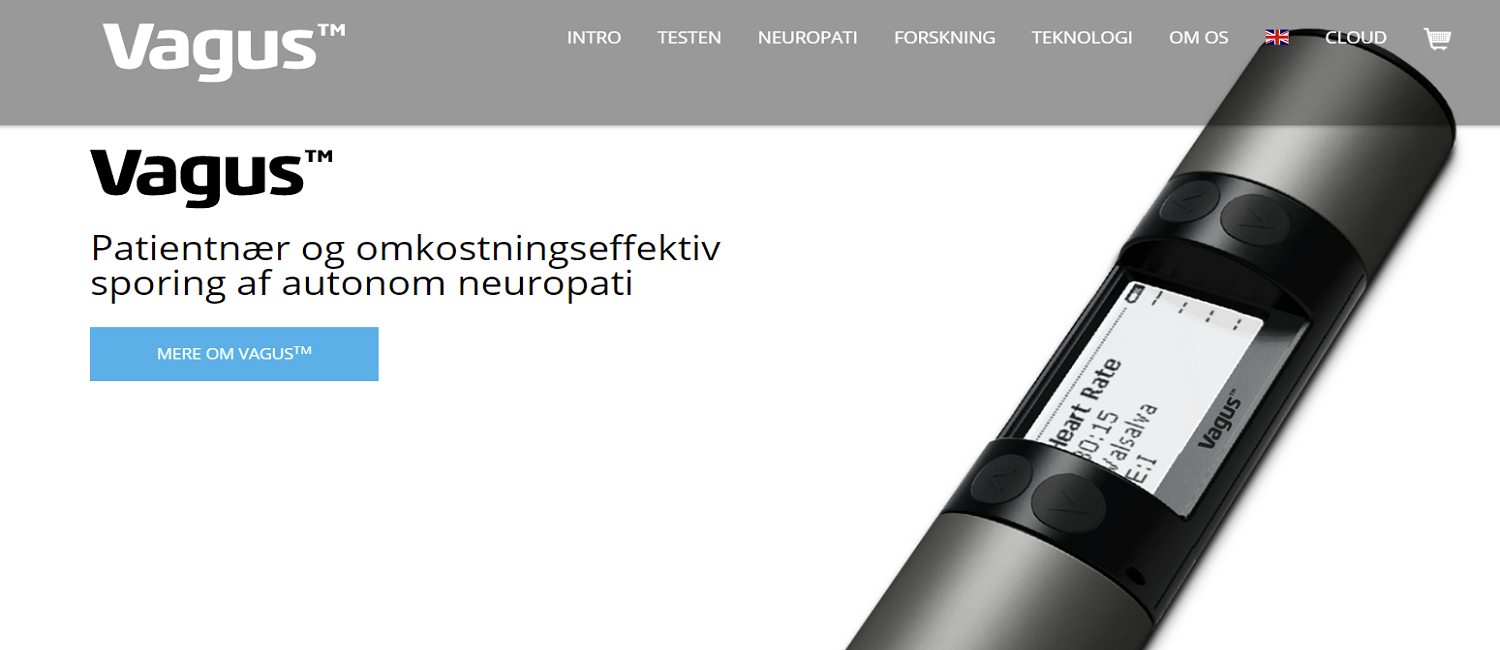
1x Vagus™ Device
2x Electrodes
2x Single Use Mouthpieces
1x Printed Manual
1x Test Pad
1x SD Memory Card
2x AA (LR6) Batteries
1x Storage Case
Cardiovascular Autonomic Neuropathy (CAN) is a common and revisable complication in diabetes, though not only in diabetes. Today, 1 in 11 adults are living with diabetes or a staggering 463 million people and 10% of global health expenditure is spent on the disease. The annual expenditures are $ 324 billion in the US and $ 161 billion in Europe. 50% of people are estimated to have CAN after ten years of diabetes. Patients with CAN have a five-fold increased risk of mortality due to a high-risk of cardiac arrhythmias, silent myocardial ischaemia and sudden death. CAN is associated with increased risk of preventable and treatable complications: hypoglycemia, blood pressure instability and sleep apnea. Screening for CAN is recommended according to international guidelines (ADA and EASD) once per year for all patient with diabetes. Different laboratory tools exist to diagnose CAN. However, these systems are advanced- and cost-intensive.
Vagus™ Ongoing production and CE marked class IIa medical diagnostic device, with single use utensils.
Vagus™ is the only device suited, and clinical validated, for the internationally recommended large scale screening of CAN. The entire test takes 5-8 minutes and is easy to fit into clinical practice. MD assistance is not required.
Vagus™ list price is 75% of the purchase price of laboratory devices. Consumables (electrodes and single-use mouthpieces) list price is 50% of laboratory devices.
Vagus™ is currently used by more than 30 hospitals in Scandinavia, UK, France.
Vagus™ has a clear pathway to FDA 510(k). Reimbursement for diagnostic Vagus tests in the US is already in place and the ROI of the Vagus device is within 3 weeks.
Vagus™ is NHS approved for UK based hospitals.
We are currently in negotiations with distribution partners for Vagus in the US – Vagus.com
40 min prediction of hypoglycemia
Cardiovascular Autonomic Neuropathy (CAN) is tied to symptomatic as well as asymptomatic hypoglycemia, which can have a significant impact on a patient's daily life.
Using the newest knowledge within AI algorithm development our invention, PreHypo, makes prediction of hypoglycemia possible in people with diabetes and CAN.
Hypoglycemia can occur if you eat less than usual after taking your regular dose of diabetes medication, or if you exercise more than you typically do.
Our invention enables patient empowerment by triggering a customize and personalized alarm from the patients or relative’s smartphone / smartwatch. This enables early intervention to avoid or reduce hypoglycemia.
PreHypo.com
Quality
MEDICUS quality management system Complies with the requirements of: ISO 13485:2016/NS-EN ISO 13485:2016
-

Quality
ISO 13485:2016/NS-EN ISO 13485:2016
Contact
Leave a Message
MEDICUS engineering ApS
Palle Juul-Jensens Boulevard 82
8200 Aarhus N
Denmark
mail@medicuseng.com